- Blog
- Cat mario 4 play
- Proofs keybase
- Barry hansen fish heads
- Mmo like ascension tv show
- Beholder lair
- Red kite info
- Revizto plus
- Into the waves necklace
- Two types of nodes occur in atomic orbitals
- Teen webcam capture gif
- Alpha omega elite carseat reviews
- Best version of logitech media server
- Foreigh currency rates
- Safari 5-1-10 download for windows
Note: Orbitals of d subshell are Equivalent in energy. (iii) ) Nodal Plane d xy xz&yz nodal plane Another type of bond, a pi (p) bond is formed when two p orbitals overlap. (ii) Nodal Surface 3 d xy 0 Nodal surface This overlap may involve s-s, s-p, s-d or even p-d orbitals. five electron cloud and can be represent as follows:Įach d-orbital of higher energy level are also double dumbell shaped but they have nodal surface. It is also formed for each bonding molecular orbital. It implies that d subshell has 5 orbitals i.e. An out-of-phase overlap forms an ANTIBONDING MOLECULAR ORBITAL. The shapes of all d- orbital is not identical. It implies that d subshell of any energy shell has five orbitals. Each p orbital of higher energy level are also dumb bell shape but they have nodal surface.Ĭase III When = 2, ‘m’ has five values -2, -1, 0, +1, +2. The two lobes of each orbital are separated by a plane of zero electron density called nodal plane.ģ. Each p orbital has dumb bell shape (2 lobes which are separated from each other by a point of zero probability called nodal point or node or nucleus).Ģ. These three p-orbitals are situated at right angle to another and are directed along x, y and z axes (figure)ġ. They, however, differ in their direction & distribution of the charge. These three p-orbitals, possesses equivalent energy and therefore, have same relation with thenucleus. It is represented in terms of n and I quantum number, N n. The total number of nodes of an orbital is the sum of the radial and angular nodes. The radial nodes are the spheres at fixed radius that occur as the principal quantum number increases while angular nodes are flat at fixed angles. We have three p-orbitals, commonly known as p x, p y and p z. The two types of nodes are: radial and angular nodes. It implies that, p subshell have three orbitals called as p x, p y and p z. The allowed values of ml 5 l, l 2 1., 2l give the number of orbitals of that type. Shapes of s-orbitals: The s-orbitals are spherically symmetrical about the nucleus (2) Angular nodes/ number of nodal planes number of angular nodes/ nodal planes =Ĭase-I : If =0 and m = 0 it implies that s subshell has only one orbital called as s orbital.

x, y, z) function which depend on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. (1) Radial nodes/ spherical nodes number of radial nodes = It has 3 possible orientation Each p orbital consists of two sections called lobes that are on either side of the plane that passes through the nucleus The. The two colors show the phase or sign of the wave function in each region.
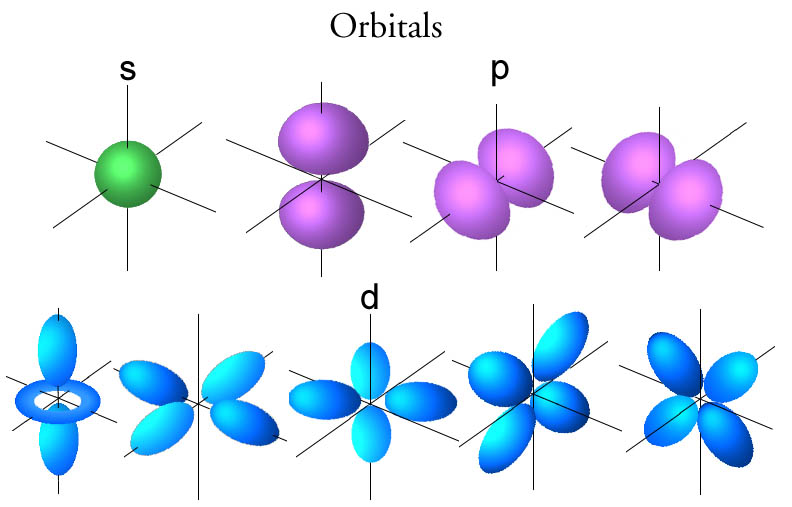
Total number of nodes = n-1 There are of 2 types. Node: It is point/ line/ plane/ surface in which probability of finding electron is zero. Types of Nodes: Nodes are of two types: a) Radial Node b) Angular Node A radial node is the spherical region around then nucleus, where the probability if.
